Industrial Ficin Meat Tenderization: Ficin Specification Checklist
Compare ficin specifications for meat tenderization: activity, pH, temperature, dosage, QC, COA/TDS/SDS, pilot validation, and cost-in-use.

For meat processors comparing protease options, industrial ficin offers a plant-derived cysteine protease route for controlled tenderization when activity, dosage, pH, temperature, and supplier documentation are specified correctly.

Why compare ficin for meat tenderization?
Ficin is a proteolytic enzyme obtained from fig latex, which is why buyers may search for ficin enzyme, fig enzyme, or ficin meat tenderizer when evaluating plant-derived proteases. In industrial ficin meat tenderization, the goal is not simply to soften meat, but to achieve repeatable texture without excessive protein breakdown, purge, or mushy bite. Compared with other proteases, ficin can be useful where processors want a cysteine protease profile for muscle and connective tissue modification, but performance depends strongly on activity units, carrier system, dispersion, contact time, pH, and heat history. A B2B specification should therefore compare ficin by measurable performance in the intended raw material: beef, pork, poultry, or further-processed products. The best purchasing decision combines enzyme potency with pilot data, documentation quality, supply reliability, and cost-in-use under the buyer’s actual process.
Use application trials to compare enzyme lots, not only catalog activity. • Define target texture with shear force, sensory, or internal bite tests. • Confirm that the enzyme format disperses in brine, marinade, or dry blend.
Core ficin specification checklist
A strong industrial ficin enzyme meat tenderization specification starts with activity method and unit definition. Protease activity can vary by assay substrate, pH, temperature, and reporting convention, so two products with similar label claims may perform differently in meat. Ask the ficin enzyme supplier for meat tenderization to state enzyme source, form, carrier, activity range, moisture, microbiological limits, heavy metal limits where applicable, recommended storage, and shelf life. The COA should match the lot received, while the TDS should describe typical use conditions and handling. The SDS should cover worker safety, dust control, and sensitization precautions for enzyme powders. If the material is used in brine injection or tumbling, request solubility or dispersibility guidance and screen size information if relevant. For comparison buying, require a retained sample and a lot-to-lot change notification process.
COA: lot-specific activity, microbiology, moisture, and relevant purity checks. • TDS: application range, storage, dosage guidance, and compatibility notes. • SDS: safe handling, PPE, dust control, and spill response information. • Supplier files: traceability, allergen statements, and regulatory suitability for the target market.
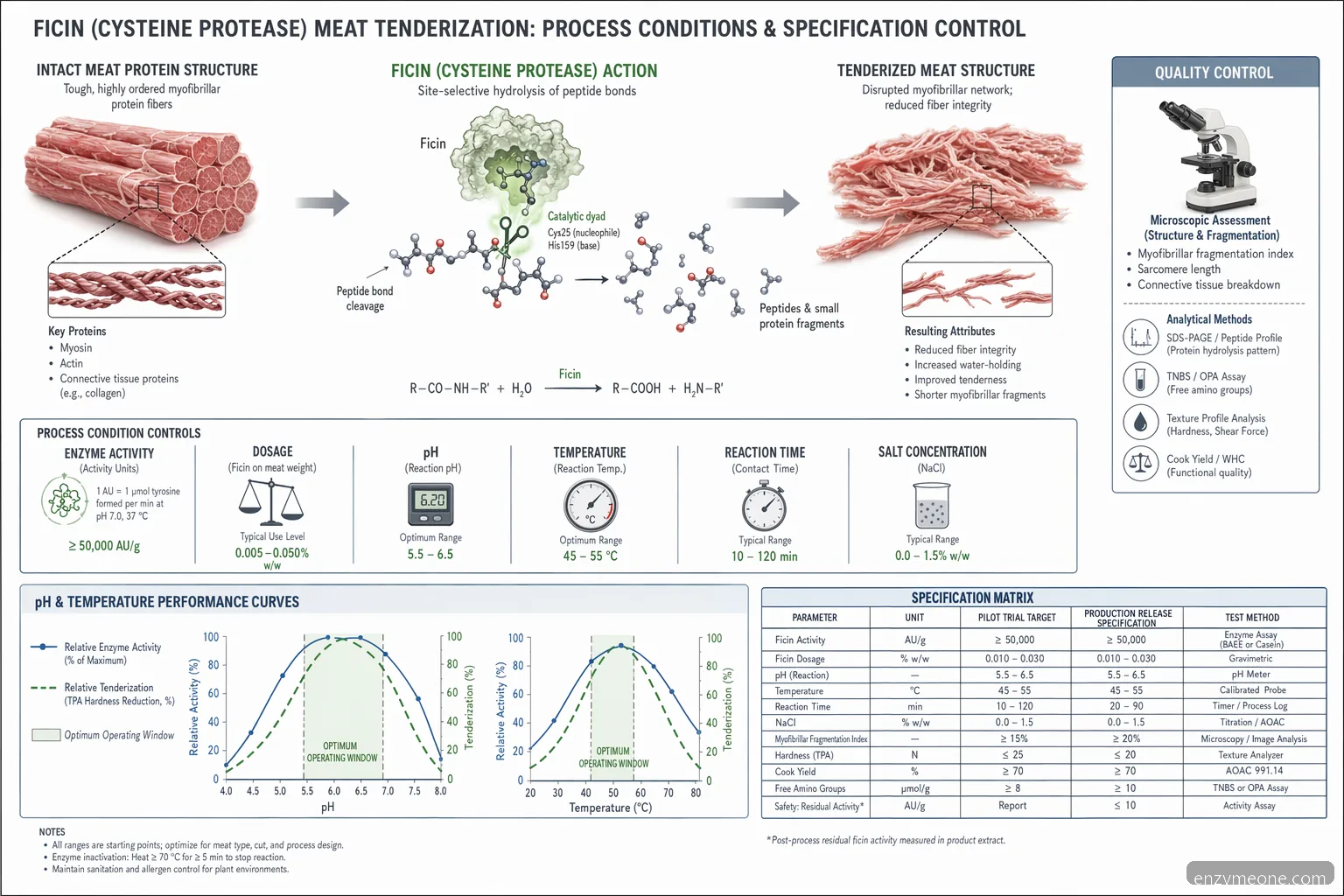
Process conditions: pH, temperature, and dosage
Ficin enzyme for meat tenderization is usually evaluated around the natural pH of meat, commonly about pH 5.4-6.2, although product-specific activity curves may cover a wider range. In marinades or brines, processors often validate pH 5.5-7.0 to balance enzyme activity, flavor system stability, and finished-product quality. Temperature is equally important: refrigerated holding at 0-4°C slows proteolysis and supports food safety programs, while activity increases during tempering, cooking ramps, or warm processing. Many ficin preparations are substantially reduced or inactivated during cooking, but the inactivation point should be validated for the specific enzyme and product geometry. Initial dosage screens often start in broad bands such as 0.02-0.20% of commercial enzyme preparation, or an activity-based equivalent, then narrow by texture and purge data. Overdosing can create soft surfaces, poor sliceability, or yield loss.
Validate dosage by activity units per kg of meat, not only percent addition. • Monitor contact time from addition through heat inactivation. • Use cold-chain controls to limit uncontrolled tenderization before cooking. • Confirm brine pH after all phosphates, salts, acids, and flavors are added.
QC checks for pilot and production trials
Before approving a ficin supplier for meat tenderization, run a structured pilot that mirrors production equipment and distribution time. For injected products, confirm brine pickup, injection uniformity, needle performance, and purge after tumbling and holding. For marinaded or tumbled products, check enzyme dispersion, surface over-tenderization, and batch-to-batch mixing time. Useful QC endpoints include raw pH, cooked yield, purge, sliceability, texture analyzer data, sensory bite panels, and visual assessment of muscle fiber integrity. If the product is cooked, record internal temperature profiles to determine when ficin activity is reduced. For raw marinated products, define the maximum refrigerated shelf life before texture drifts outside specification. Industrial fig enzyme meat tenderization trials should include a no-enzyme control and at least two dosage levels. This prevents confusing enzyme performance with raw material variability, aging, salt level, or mechanical tenderization effects.
Run no-enzyme, low-dose, target-dose, and high-dose comparisons. • Track raw material age, cut, fat level, and supplier source. • Measure purge after hold and after thermal processing. • Retain photos and cooked samples for cross-functional review.
Comparing ficin with other protease options
Comparison intent matters because ficin is rarely evaluated in isolation. Meat processors may compare ficin with papain, bromelain, microbial proteases, or blended systems. Ficin may be selected for a specific tenderization profile, plant enzyme positioning, or performance in a defined pH and temperature window. However, a different protease may be more economical if it reaches the target texture at a lower cost-in-use or with less risk of over-tenderization. The practical comparison should use equal finished-product targets rather than equal grams of enzyme. Standardize meat block, brine, pH, salt, tumbling energy, hold time, and cook schedule. Then compare yield, texture, sensory acceptance, rework risk, labeling requirements, and operational simplicity. Industrial ficin protein hydrolysis experience may also be relevant if the supplier understands controlled proteolysis, but meat tenderization needs its own validation because intact muscle behaves differently from slurry hydrolysis.
Compare on cost per accepted kg of finished product. • Include rework, purge, and rejected texture in the cost model. • Avoid switching enzymes without revalidating time-temperature controls.
Supplier qualification and cost-in-use
A reliable ficin enzyme supplier for meat tenderization should support technical, quality, and procurement teams. Beyond price per kilogram, request lot consistency data, lead time, minimum order quantity, packaging options, storage conditions, and documentation turnaround. Supplier qualification should include COA/TDS/SDS review, sample approval, pilot validation, and a written specification that identifies the activity method and acceptance range. If the enzyme is imported or used in regulated food markets, confirm that the supplier can provide documentation appropriate to the destination and application without relying on vague claims. Cost-in-use should calculate dosage by enzyme activity, finished yield, labor impact, process time, purge reduction or increase, complaint risk, and the value of consistent tenderness. A higher-activity ficin can be cheaper in use if it reduces dosage and shipping, but only when dispersion and performance remain consistent in the plant.
Qualify at least one backup lot or backup supplier where supply risk is critical. • Set incoming QC checks for identity, activity, appearance, and documentation. • Review change-control expectations before commercial approval.
Technical Buying Checklist
Buyer Questions
Ficin is a fig-derived cysteine protease that can hydrolyze meat proteins under controlled conditions. In industrial ficin meat tenderization, it is valued when processors need measurable tenderization through brine, marinade, tumbling, or formulated systems. Its usefulness depends on activity, dosage, contact time, pH, temperature, and cook-step control. It should be validated against texture, purge, yield, and sensory targets before commercial approval.
A practical first screen is often a broad range such as 0.02-0.20% of the commercial ficin preparation, but the final dosage should be based on enzyme activity units per kg of meat. The right level depends on cut, raw material age, pH, salt, injection pickup, hold time, and heat process. Always include a no-enzyme control and evaluate purge, texture, sliceability, and eating quality.
A qualified ficin supplier for meat tenderization should provide a lot-specific COA, current TDS, and SDS. Buyers should also request traceability, storage and shelf-life guidance, application recommendations, and regulatory suitability statements for the intended market. The COA should clearly state activity and test method. The TDS should describe typical pH, temperature, dosage, and handling guidance without replacing plant-level validation.
Compare ficin, papain, bromelain, or microbial proteases under the same meat block, brine, pH, dosage basis, contact time, and cook schedule. Do not compare only price per kg or label activity. Compare finished-product texture, yield, purge, sensory quality, process robustness, documentation, and cost per accepted kg. Ficin may be preferred in some systems, while another protease may be better in others.
Industrial ficin protein hydrolysis and industrial ficin pharmaceutical applications may use related protease functionality, but they require different specifications, controls, and validation. Meat tenderization focuses on intact muscle texture, yield, and food processing performance. Protein hydrolysis may focus on degree of hydrolysis, peptide profile, and bitterness. Pharmaceutical use requires application-specific quality and regulatory review beyond a standard food-processing tenderizer specification.
Related Search Themes
industrial ficin enzyme meat tenderization, industrial fig enzyme meat tenderization, ficin supplier for meat tenderization, ficin for meat tenderization, ficin enzyme supplier for meat tenderization, ficin enzyme for meat tenderization
Ficin for Research & Industry
Need Ficin for your lab or production process?
ISO 9001 certified · Food-grade & research-grade · Ships to 80+ countries
Frequently Asked Questions
What makes ficin useful for industrial meat tenderization?
Ficin is a fig-derived cysteine protease that can hydrolyze meat proteins under controlled conditions. In industrial ficin meat tenderization, it is valued when processors need measurable tenderization through brine, marinade, tumbling, or formulated systems. Its usefulness depends on activity, dosage, contact time, pH, temperature, and cook-step control. It should be validated against texture, purge, yield, and sensory targets before commercial approval.
What dosage of ficin should a meat processor start with?
A practical first screen is often a broad range such as 0.02-0.20% of the commercial ficin preparation, but the final dosage should be based on enzyme activity units per kg of meat. The right level depends on cut, raw material age, pH, salt, injection pickup, hold time, and heat process. Always include a no-enzyme control and evaluate purge, texture, sliceability, and eating quality.
What documents should a ficin supplier provide?
A qualified ficin supplier for meat tenderization should provide a lot-specific COA, current TDS, and SDS. Buyers should also request traceability, storage and shelf-life guidance, application recommendations, and regulatory suitability statements for the intended market. The COA should clearly state activity and test method. The TDS should describe typical pH, temperature, dosage, and handling guidance without replacing plant-level validation.
How should ficin be compared with papain or bromelain?
Compare ficin, papain, bromelain, or microbial proteases under the same meat block, brine, pH, dosage basis, contact time, and cook schedule. Do not compare only price per kg or label activity. Compare finished-product texture, yield, purge, sensory quality, process robustness, documentation, and cost per accepted kg. Ficin may be preferred in some systems, while another protease may be better in others.
Can ficin be used for protein hydrolysis or pharmaceutical applications?
Industrial ficin protein hydrolysis and industrial ficin pharmaceutical applications may use related protease functionality, but they require different specifications, controls, and validation. Meat tenderization focuses on intact muscle texture, yield, and food processing performance. Protein hydrolysis may focus on degree of hydrolysis, peptide profile, and bitterness. Pharmaceutical use requires application-specific quality and regulatory review beyond a standard food-processing tenderizer specification.
Related: Ficin Enzyme Uses for Industrial Proteolysis
Turn This Guide Into a Supplier Brief Request a ficin specification review, pilot sample, and cost-in-use comparison for your meat tenderization process. See our application page for Ficin Enzyme Uses for Industrial Proteolysis at /applications/ficin-enzyme-uses/ for specs, MOQ, and a free 50 g sample.
Contact Us to Contribute